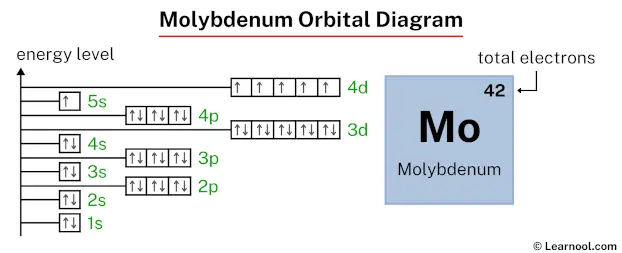
In the molybdenum orbital diagram, the 1s subshell holds two electrons, the 2s subshell carries another pair, the 2p subshell encompasses six electrons, the 3s subshell contains two electrons, the 3p subshell carries six electrons, the 4s subshell holds two electrons, the 3d subshell carries ten electrons, the 4p subshell accommodates six electrons, the 5s subshell has one electron, and the 4d subshell carries five electrons, totaling forty-two electrons.
To illustrate the molybdenum orbital diagram, begin by determining the number of electrons from the periodic table. Take note of the electron configuration for reference and follow the three fundamental rules: the Aufbau principle, Pauli exclusion principle, and Hund’s rule. This systematic approach ensures an accurate representation of molybdenum’s orbital arrangement.
Steps
Find electrons
The atomic number of molybdenum represents the total number of electrons of molybdenum. Since the atomic number of molybdenum is 42, the total electrons of molybdenum are 42.
Write electron configuration
The electron configuration of molybdenum is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d5.
Learn how to find: Molybdenum electron configuration
Now in the next step, start drawing the orbital diagram for molybdenum.
Draw orbital diagram
Before drawing the orbital diagram, you should know the three general rules.
- Aufbau principle – electrons are first filled in lowest energy orbital and then in higher energy orbital
- Pauli exclusion principle – two electrons with the same spin can not occupy the same orbital
- Hund’s rule – each orbital should be first filled with one electron before being paired with a second electron
Also, you should know the number of orbitals in each subshell.
We can calculate the number of orbitals in each subshell using the formula: 2ℓ + 1
Where, ℓ = azimuthal quantum number of the subshell
For s subshell, ℓ = 0
For p subshell, ℓ = 1
For d subshell, ℓ = 2
For f subshell, ℓ = 3
So each s subshell has one orbital, each p subshell has three orbitals, each d subshell has five orbitals, and each f subshell has seven orbitals.
Now start to draw!
As mentioned above, the electron configuration of molybdenum is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d5. Hence, draw the blank orbital diagram of molybdenum up to 4d subshell as follows:

In the above orbital diagram, the box represents an orbital. Each orbital has a capacity of two electrons. And the arrows (↑↓) are drawn inside the box to represent electrons.
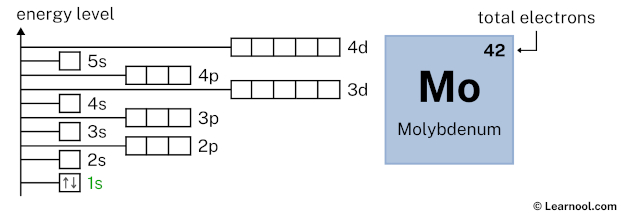
Now 1s2 indicates that the 1s subshell has 2 electrons. So draw two arrows in the 1s box showing two electrons as follows:
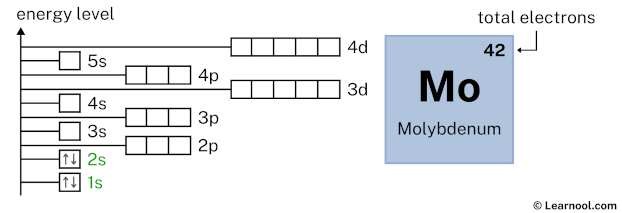
2s2 indicates that the 2s subshell has 2 electrons. So draw two arrows in the 2s box showing two electrons as follows:
2p6 indicates that the 2p subshell has 6 electrons. So draw six arrows in the 2p box showing six electrons as follows:
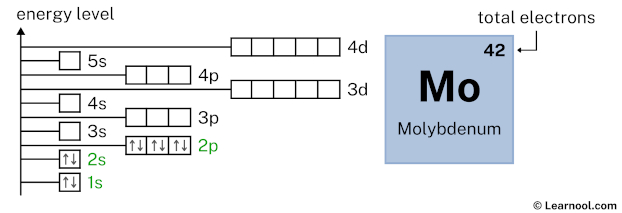
3s2 indicates that the 3s subshell has 2 electrons. So draw two arrows in the 3s box showing two electrons as follows:

3p6 indicates that the 3p subshell has 6 electrons. So draw six arrows in the 3p box showing six electrons as follows:

4s2 indicates that the 4s subshell has 2 electrons. So draw two arrows in the 4s box showing two electrons as follows:
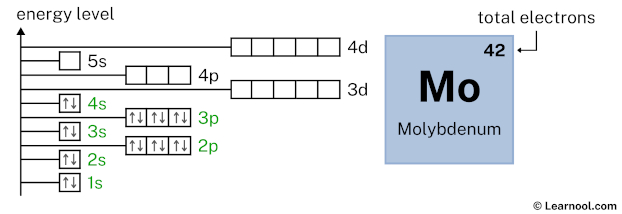
3d10 indicates that the 3d subshell has 10 electrons. So draw ten arrows in the 3d box showing ten electrons as follows:
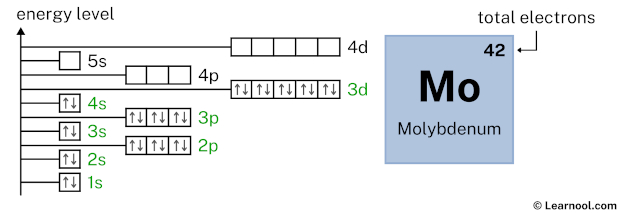
4p6 indicates that the 4p subshell has 6 electrons. So draw six arrows in the 4p box showing six electrons as follows:
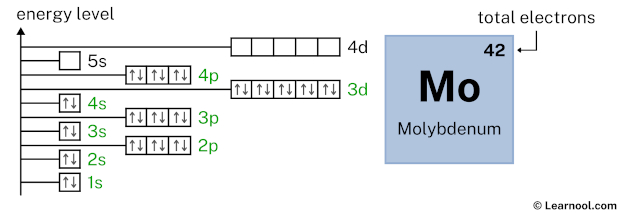
5s1 indicates that the 5s subshell has 1 electron. So draw one arrow in the 5s box showing one electron as follows:

4d5 indicates that the 4d subshell has 5 electrons. So draw five arrows in the 4d box showing five electrons as follows:
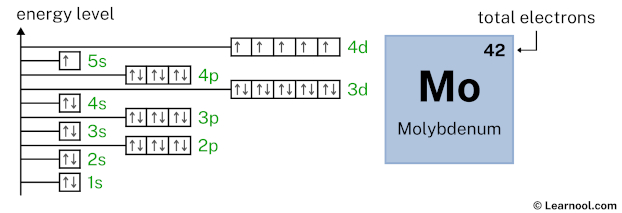
That’s it! This is the final orbital diagram of molybdenum as we have used all 42 electrons.
Question: Why does the 5s subshell have only one electron (instead of two electrons), and the 4d subshell has five electrons (instead of four electrons)?
Answer: A half-full 4d subshell is more stable than a partially filled 4d subshell. That’s why the 4d subshell has five electrons (instead of four electrons). And the 5s subshell has one electron (instead of two electrons).
Next: Tantalum orbital diagram
Related
More topics
- Molybdenum
- Molybdenum Bohr model
- Molybdenum electron configuration
- Molybdenum protons neutrons electrons
External links
- https://www.numerade.com/ask/question/give-a-shorthand-orbital-diagram-for-molybdenum-12093/
- https://www.chegg.com/homework-help/questions-and-answers/-following-correct-orbital-diagram-ground-state-electron-configurastion-molybdenum-mo-z-42-q33102767
Deep
Learnool.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
