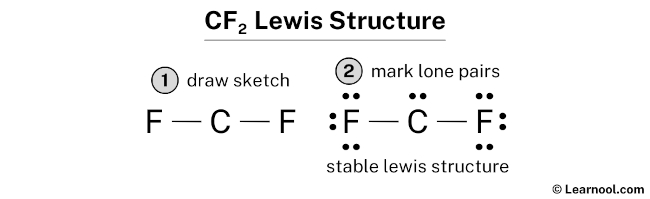
CF2 (difluorocarbene) has one carbon atom and two fluorine atoms.
In CF2 Lewis structure, there are two single bonds around the carbon atom, with two fluorine atoms attached to it. Each fluorine atom has three lone pairs, and the carbon atom has one lone pair.
Alternative method: Lewis structure of CF2
Rough sketch
- First, determine the total number of valence electrons
In the periodic table, carbon lies in group 14, and fluorine lies in group 17.
Hence, carbon has four valence electrons and fluorine has seven valence electrons.
Since CF2 has one carbon atom and two fluorine atoms, so…
Valence electrons of one carbon atom = 4 × 1 = 4
Valence electrons of two fluorine atoms = 7 × 2 = 14
And the total valence electrons = 4 + 14 = 18
Learn how to find: Carbon valence electrons and Fluorine valence electrons
- Second, find the total electron pairs
We have a total of 18 valence electrons. And when we divide this value by two, we get the value of total electron pairs.
Total electron pairs = total valence electrons ÷ 2
So the total electron pairs = 18 ÷ 2 = 9
- Third, determine the central atom
We have to place the least electronegative atom at the center.
Since carbon is less electronegative than fluorine, assume that the central atom is carbon.
Therefore, place carbon in the center and fluorines on either side.
- And finally, draw the rough sketch
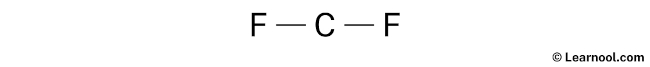
Lone pair
Here, we have a total of 9 electron pairs. And two C — F bonds are already marked. So we have to only mark the remaining seven electron pairs as lone pairs on the sketch.
Also remember that both (carbon and fluorine) are the period 2 elements, so they can not keep more than 8 electrons in their last shell.
Always start to mark the lone pairs from outside atoms. Here, the outside atoms are fluorines.
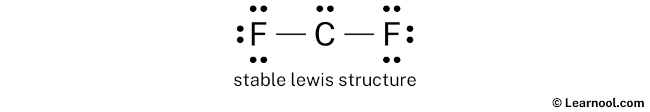
So for each fluorine, there are three lone pairs, and for carbon, there is one lone pair.
Mark the lone pairs on the sketch as follows:
Formal charge
Use the following formula to calculate the formal charges on atoms:
Formal charge = valence electrons – nonbonding electrons – ½ bonding electrons
For carbon atom, formal charge = 4 – 2 – ½ (4) = 0
For each fluorine atom, formal charge = 7 – 6 – ½ (2) = 0
Here, both carbon and fluorine atoms do not have charges, so no need to mark the charges.
Final structure
The final structure of CF2 contains a central carbon atom connected to two fluorine atoms through single covalent bonds. Within this arrangement, the carbon atom retains one lone pair and forms two bonds, which leaves it with a sextet of electrons—a common occurrence for highly reactive carbene molecules. Each fluorine atom fulfills its octet by maintaining three lone pairs of its own alongside the single shared bond. This setup is the most stable for the singlet state because it results in formal charges of zero for all atoms, representing the most energetically favorable state for the molecule. Hence, this specific electronic distribution serves as the definitive and most accurate Lewis representation of difluorocarbene.
Next: SeO Lewis structure
External links
- https://www.chegg.com/homework-help/questions-and-answers/cf2-difluoroacetylene-lewis-structure-use-fc-cf-chain-arrangement-sketch-3d-shape-bond-ang-q54740766
- https://www.numerade.com/ask/question/which-of-the-four-following-lewis-structures-is-expected-to-be-the-dominant-lewis-structure-for-the-cf2-molecule-26983/
- https://www.coursehero.com/tutors-problems/Chemistry/27699407-Lewis-Structures/
Deep
Learnool.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
