In the silver Bohr model, the nucleus holds 47 protons and 61 neutrons. Encircling this nucleus are five electron shells, carrying a total of 47 electrons.
To draw the silver Bohr model, represent the 47 protons, 61 neutrons, and 47 electrons. Begin by sketching the nucleus, and then draw the five electron shells. The first four shells should contain 2, 8, 18, and 18 electrons, respectively, while the fifth shell holds the remaining 1 electron.
Steps
Write protons, neutrons, and electrons of silver atom
Silver has 47 protons, 61 neutrons, and 47 electrons.
Learn how to find: Silver protons neutrons electrons
Draw nucleus of silver atom
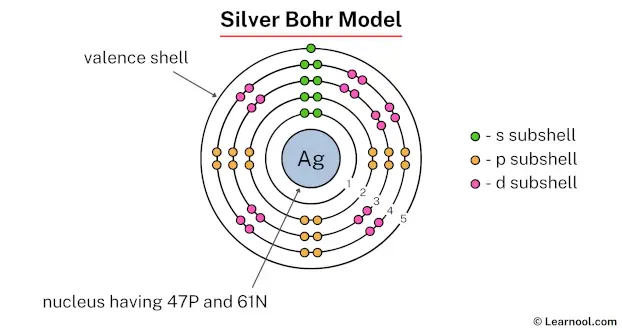
The nucleus of a silver atom contains 47 protons and 61 neutrons. So draw the nucleus of silver atom as follows:
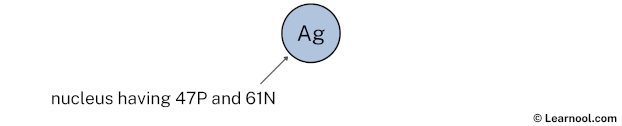
Now in the next step, draw the 1st electron shell and start marking electrons.
Draw 1st electron shell
Remember that we have a total of 47 electrons.
The 1st electron shell (containing s subshell) can hold up to a maximum of 2 electrons. So draw the 1st electron shell as follows:
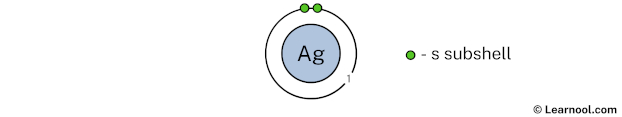
In the above image, 1 represents the 1st electron shell that contains 1s subshell. And the green color represents the number of electrons in that subshell. This means that the 1st electron shell has a total of 2 electrons.
Since we have already used 2 electrons in the 1st electron shell, now we have 47 – 2 = 45 electrons left. So in the next step, we have to draw the 2nd electron shell.
Draw 2nd electron shell
The 2nd electron shell (containing s subshell and p subshell) can hold up to a maximum of 8 electrons. So draw the 2nd electron shell as follows:
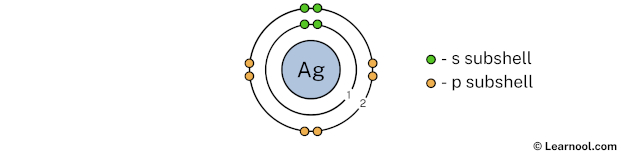
In the above image, 2 represents the 2nd electron shell that contains 2s and 2p subshells. And the green and orange color represents the number of electrons in that subshell. This means that the 2nd electron shell has a total of 8 electrons.
Now we have already used 10 electrons in 1st and 2nd electron shells, so we have 47 – 10 = 37 electrons left. So in the next step, we have to draw the 3rd electron shell.
Draw 3rd electron shell
The 3rd electron shell (containing s subshell, p subshell, and d subshell) can hold up to a maximum of 18 electrons. So draw the 3rd electron shell as follows:
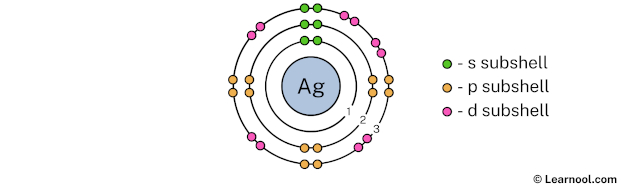
In the above image, 3 represents the 3rd electron shell that contains 3s, 3p, and 3d subshells. And the green, orange, and pink color represents the number of electrons in that subshell. This means that the 3rd electron shell has a total of 18 electrons.
Now we have already used 28 electrons in 1st, 2nd, and 3rd electron shells, so we have 47 – 28 = 19 electrons left. So in the next step, we have to draw the 4th electron shell.
Draw 4th electron shell
The 4th electron shell (containing s subshell, p subshell, d subshell, and f subshell) can hold up to a maximum of 32 electrons. So draw the 4th electron shell as follows:
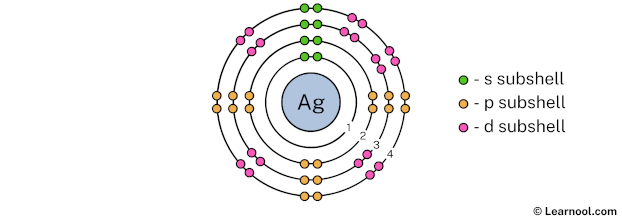
In the above image, 4 represents the 4th electron shell that contains 4s, 4p, and 4d subshells. And the green, orange, and pink color represents the number of electrons in that subshell. This means that the 4th electron shell has a total of 18 electrons.
The 4th electron shell contains only 4s, 4p, and 4d subshells, and not a 4f subshell. This is because according to the aufbau principle, the 5s subshell is filled first and then 4d, 5p, 6s… and so on.
Now we have already used 46 electrons in 1st, 2nd, 3rd, and 4th electron shells, so we have 47 – 46 = 1 electron left. So in the next step, we have to draw the 5th electron shell.
Draw 5th electron shell
The 5th electron shell can hold up to a maximum of 50 electrons. So draw the 5th electron shell as follows:
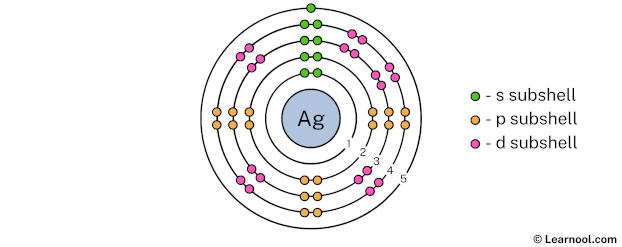
In the above image, 5 represents the 5th electron shell that contains 5s subshell. And the green color represents the number of electrons in that subshell. This means that the 5th electron shell has a total of 1 electron.
That’s it! This is the final Bohr model of silver atom as we have used all 47 electrons: 2 electrons in the 1st electron shell, 8 electrons in the 2nd electron shell, 18 electrons in the 3rd electron shell, 18 electrons in the 4th electron shell, and 1 electron in the 5th electron shell.
Question: Why does the 5s subshell have only one electron (instead of two electrons), and the 4d subshell has ten electrons (instead of nine electrons)?
Let’s draw the orbital diagram of silver, and find the answer to the above question.
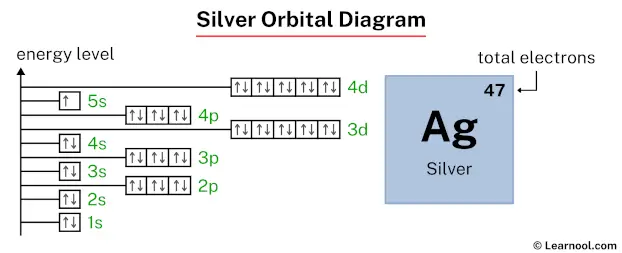
Answer: A completely full 4d subshell is more stable than a partially filled 4d subshell. That’s why the 4d subshell has ten electrons (instead of nine electrons). And the 5s subshell has one electron (instead of two electrons).
Next: Cadmium Bohr model
Related
More topics
External links
- File:47 silver (Ag) enhanced Bohr model.png – Wikimedia Commons
- Chemical Elements.com – Silver (Ag) – Chemical Elements.com
- Silver (Ag) – Periodic Table – ChemicalAid
- Silver Bohr Model – How to draw Bohr diagram for Silver (Ag)? – Topblogtenz
Deep
Learnool.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
