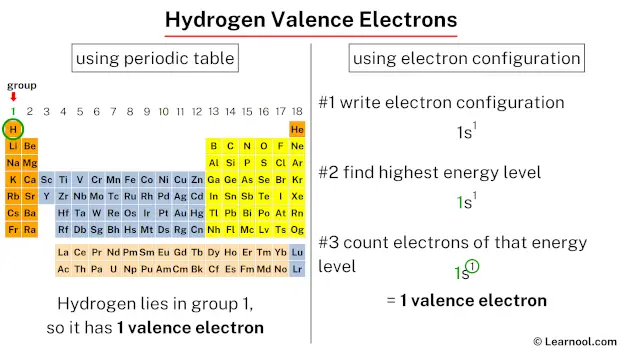
Hydrogen possesses 1 valence electron. The valence electrons of hydrogen can be determined using two different methods – by referring to the periodic table or by utilizing its electron configuration.
Methods
Using periodic table

Get the periodic table having the chemical elements marked on it as mentioned above.
Now mark the location of hydrogen on the periodic table.

Next, mark the group number of hydrogen on the periodic table.
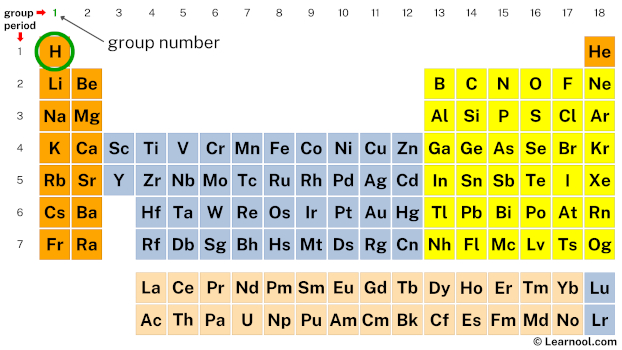
The valence electrons of each main-group element can be determined by the column in which it is located.
(i.e., all group 1 elements have 1 valence electron, all group 2 elements have 2 valence electrons, skip the transition metals… then, all group 13 elements have 3 valence electrons, all group 14 elements have 4 valence electrons, and so on up to group 18 elements)
Since hydrogen is in group 1, it has 1 valence electron.
Using electron configuration
- First, write electron configuration of hydrogen
The electron configuration of hydrogen is 1s1.
- Second, find highest energy level in electron configuration

In the above electron configuration, the highest energy level (1) is marked with green color.
- Finally, count electrons of that energy level

The 1st energy level contains 1s subshell and it has 1 electron. So hydrogen has a total of 1 valence electron.
Next: Helium valence electrons
Related
More topics
External links
- https://socratic.org/questions/how-many-valence-electrons-does-hydrogen-have
- https://homework.study.com/explanation/how-many-valence-electrons-does-hydrogen-have.html
Deep
Learnool.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
