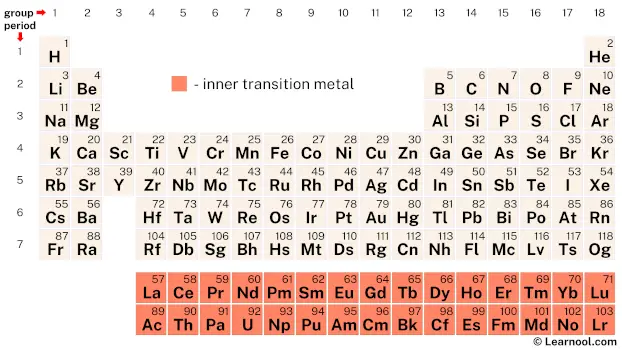
Inner transition metals, also known as f-block elements, are two series of chemical elements located within the periodic table. The first series is known as the lanthanides and consists of elements with atomic numbers ranging from 57 to 71. The second series is called the actinides and encompasses elements with atomic numbers ranging from 89 to 103. These elements are referred to as f-block elements because their last electrons enter into the f orbitals. This unique electron configuration distinguishes them from other elements on the periodic table.
Inner transition metals have a general electron configuration that involves filling the f orbitals. This leads to an incomplete outermost shell, which contributes to their characteristic chemical behavior. All inner transition metals are metals, as they possess typical metallic properties such as conductivity and malleability. For example, cerium (Ce) and uranium (U) are two inner transition metals that find practical applications. Cerium is used in catalytic converters, glass manufacturing, and as a component in alloys, while uranium is utilized as a fuel in nuclear reactors.
Inner transition metals are relatively rare in the Earth’s crust in terms of abundance. Their unstable nature, along with limited occurrence in accessible deposits, contributes to their scarcity. It’s worth noting that some inner transition metals are synthetic, meaning they do not occur naturally and must be produced through artificial processes. For instance, elements like americium (Am) through lawrencium (Lr) are synthetic and are created in nuclear reactors or particle accelerators for research and practical purposes.
List
| Lanthanum | La | 57 |
| Cerium | Ce | 58 |
| Praseodymium | Pr | 59 |
| Neodymium | Nd | 60 |
| Promethium | Pm | 61 |
| Samarium | Sm | 62 |
| Europium | Eu | 63 |
| Gadolinium | Gd | 64 |
| Terbium | Tb | 65 |
| Dysprosium | Dy | 66 |
| Holmium | Ho | 67 |
| Erbium | Er | 68 |
| Thulium | Tm | 69 |
| Ytterbium | Yb | 70 |
| Lutetium | Lu | 71 |
| Actinium | Ac | 89 |
| Thorium | Th | 90 |
| Protactinium | Pa | 91 |
| Uranium | U | 92 |
| Neptunium | Np | 93 |
| Plutonium | Pu | 94 |
| Americium | Am | 95 |
| Curium | Cm | 96 |
| Berkelium | Bk | 97 |
| Californium | Cf | 98 |
| Einsteinium | Es | 99 |
| Fermium | Fm | 100 |
| Mendelevium | Md | 101 |
| Nobelium | No | 102 |
| Lawrencium | Lr | 103 |
On periodic table
| group | ⇨ | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| period | ⇩ | ||||||||||||||||||
| 1 | 1 H  Hydrogen |
2 He  Helium |
|||||||||||||||||
| 2 | 3 Li  Lithium |
4 Be  Beryllium |
5 B  Boron |
6 C  Carbon |
7 N  Nitrogen |
8 O  Oxygen |
9 F  Fluorine |
10 Ne  Neon |
|||||||||||
| 3 | 11 Na  Sodium |
12 Mg  Magnesium |
13 Al  Aluminium |
14 Si Silicon |
15 P  Phosphorus |
16 S  Sulfur |
17 Cl  Chlorine |
18 Ar  Argon |
|||||||||||
| 4 | 19 K  Potassium |
20 Ca  Calcium |
21 Sc  Scandium |
22 Ti  Titanium |
23 V  Vanadium |
24 Cr  Chromium |
25 Mn  Manganese |
26 Fe  Iron |
27 Co  Cobalt |
28 Ni  Nickel |
29 Cu  Copper |
30 Zn  Zinc |
31 Ga  Gallium |
32 Ge  Germanium |
33 As  Arsenic |
34 Se  Selenium |
35 Br  Bromine |
36 Kr  Krypton |
|
| 5 | 37 Rb  Rubidium |
38 Sr  Strontium |
39 Y  Yttrium |
40 Zr  Zirconium |
41 Nb  Niobium |
42 Mo  Molybdenum |
43 Tc  Technetium |
44 Ru  Ruthenium |
45 Rh  Rhodium |
46 Pd  Palladium |
47 Ag  Silver |
48 Cd  Cadmium |
49 In  Indium |
50 Sn  Tin |
51 Sb  Antimony |
52 Te  Tellurium |
53 I  Iodine |
54 Xe  Xenon |
|
| 6 | 55 Cs  Caesium |
56 Ba  Barium |
72 Hf  Hafnium |
73 Ta  Tantalum |
74 W  Tungsten |
75 Re  Rhenium |
76 Os  Osmium |
77 Ir  Iridium |
78 Pt  Platinum |
79 Au  Gold |
80 Hg  Mercury |
81 Tl  Thallium |
82 Pb  Lead |
83 Bi  Bismuth |
84 Po  Polonium |
85 At  Astatine |
86 Rn  Radon |
||
| 7 | 87 Fr  Francium |
88 Ra  Radium |
104 Rf  Rutherfordium |
105 Db  Dubnium |
106 Sg  Seaborgium |
107 Bh  Bohrium |
108 Hs  Hassium |
109 Mt  Meitnerium |
110 Ds  Darmstadtium |
111 Rg  Roentgenium |
112 Cn  Copernicium |
113 Nh  Nihonium |
114 Fl  Flerovium |
115 Mc  Moscovium |
116 Lv  Livermorium |
117 Ts  Tennessine |
118 Og  Oganesson |
||
| 57 La  Lanthanum |
58 Ce  Cerium |
59 Pr  Praseodymium |
60 Nd  Neodymium |
61 Pm  Promethium |
62 Sm  Samarium |
63 Eu  Europium |
64 Gd  Gadolinium |
65 Tb  Terbium |
66 Dy  Dysprosium |
67 Ho  Holmium |
68 Er  Erbium |
69 Tm  Thulium |
70 Yb  Ytterbium |
71 Lu  Lutetium |
|||||
| 89 Ac  Actinium |
90 Th  Thorium |
91 Pa  Protactinium |
92 U  Uranium |
93 Np  Neptunium |
94 Pu  Plutonium |
95 Am  Americium |
96 Cm  Curium |
97 Bk  Berkelium |
98 Cf  Californium |
99 Es  Einsteinium |
100 Fm  Fermium |
101 Md  Mendelevium |
102 No  Nobelium |
103 Lr  Lawrencium |
|||||
| – inner transition metal |
On the periodic table, the inner transition metals include the lanthanides – elements with atomic numbers ranging from 57 to 71 – and the actinides – elements with atomic numbers ranging from 89 to 103.
Related
More topics
- Transuranium element
- Superheavy element
- Rare-earth element
- Synthetic element
- Main-group element
- Inner transition metal
External links
- Inner transition metal – Wikipedia
- Inner Transition Metals – Angelo State University
- Periodic Table of the Elements – Inner Transition Metals – Cool Periodic Table
- Inner Transition Metals – Nuclear Chemistry – Wikibooks
- Inner transition element – New World Encyclopedia
- Transition Metals – Purdue University
- Inner transition metal Facts for Kids – Kids encyclopedia facts
- Transition metal | Definition, Properties, Elements, & Facts – Britannica
- 16 Enigmatic Facts About Inner Transition Metal – Facts.net
- Inner Transition Metals – Concept – Chemistry Video – Brightstorm
- Periodic Table – An Introduction to Chemistry
- What are the elements classified as inner transition metals? – CK-12 Foundation
- Inner Transition Metals of the Periodic Table – Pediabay
- Are inner transition metals also part of the transition group? – Chemistry Stack Exchange
- What are inner transition metals? – Homework.Study.com
- Inner Transition Metals – Science Struck
- What is the usage of inner transition metals? – Quora
- Where are Inner Transition Metals located on Periodic Table? – Periodic Table Guide
- Explain the meaning of inner transition series? – Socratic
Deep
Learnool.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
